Characteristics A layer of solid xenon floating on top of liquid xenon inside a high voltage apparatus. It was the first time atoms had been precisely positioned on a flat surface. The program, called IBM in atoms, used a scanning tunneling microscope to arrange 35 individual xenon atoms on a substrate of chilled crystal of nickel to spell out the three letter company initialism. In November 1989, IBM scientists demonstrated a technology capable of manipulating individual atoms. By 1971, more than 80 xenon compounds were known. 
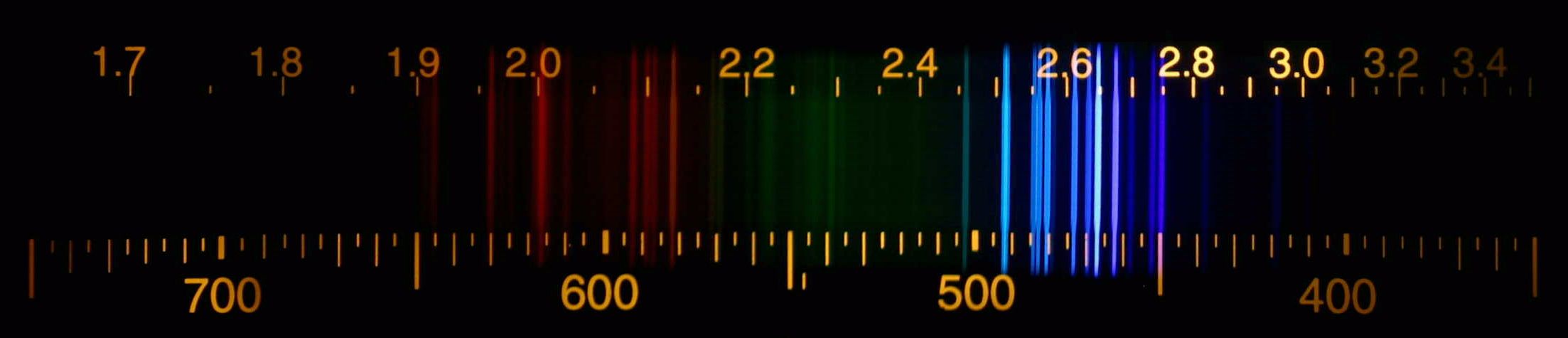
Since then, many other xenon compounds have been discovered, in addition to some compounds of the noble gases argon, krypton, and radon, including argon fluorohydride (HArF), krypton difluoride (KrF 2), and radon fluoride. īartlett thought its composition to be Xe + −, but later work revealed that it was probably a mixture of various xenon-containing salts. On March 23, 1962, he mixed the two gases and produced the first known compound of a noble gas, xenon hexafluoroplatinate. Since O 2(1165 kJ/mol) and xenon (1170 kJ/mol) have almost the same first ionization potential, Bartlett realized that platinum hexafluoride might also be able to oxidize xenon. However, while teaching at the University of British Columbia, Neil Bartlett discovered that the gas platinum hexafluoride (PtF 6) was a powerful oxidizing agent that could oxidize oxygen gas (O 2) to form dioxygenyl hexafluoroplatinate ( O + Xenon and the other noble gases were for a long time considered to be completely chemically inert and not able to form compounds. An acrylic cube specially prepared for element collectors containing liquefied xenon Cullen, who successfully used it with two patients. Xenon was first used as a surgical anesthetic in 1951 by American anesthesiologist Stuart C. Lazarev apparently studied xenon anesthesia in 1941, the first published report confirming xenon anesthesia was in 1946 by American medical researcher John H. From his results, he deduced that xenon gas could serve as an anesthetic. He tested the effects of varying the breathing mixtures on his subjects, and discovered that this caused the divers to perceive a change in depth. began exploring the causes of "drunkenness" in deep-sea divers. In 1934, Edgerton was able to generate flashes as brief as one microsecond with this method. This led him to the invention of the xenon flash lamp in which light is generated by passing brief electric current through a tube filled with xenon gas. ĭuring the 1930s, American engineer Harold Edgerton began exploring strobe light technology for high speed photography. In 1902, Ramsay estimated the proportion of xenon in the Earth's atmosphere to be one part in 20 million. Ramsay suggested the name xenon for this gas from the Greek word ξένον xénon, neuter singular form of ξένος xénos, meaning 'foreign(er)', 'strange(r)', or 'guest'. They found xenon in the residue left over from evaporating components of liquid air. Xenon was discovered in England by the Scottish chemist William Ramsay and English chemist Morris Travers in September 1898, shortly after their discovery of the elements krypton and neon. Radioactive xenon-135 is produced by beta decay from iodine-135 (a product of nuclear fission), and is the most significant (and unwanted) neutron absorber in nuclear reactors. More than 40 unstable xenon isotopes undergo radioactive decay, and the isotope ratios of xenon are an important tool for studying the early history of the Solar System. Naturally occurring xenon consists of seven stable isotopes and two long-lived radioactive isotopes. Xenon is also used to search for hypothetical weakly interacting massive particles Īnd as a propellant for ion thrusters in spacecraft. The first excimer laser design used a xenon dimer molecule (Xe 2) as the lasing medium, and the earliest laser designs used xenon flash lamps as pumps. Xenon is used in flash lamps and arc lamps, and as a general anesthetic. Although generally unreactive, it can undergo a few chemical reactions such as the formation of xenon hexafluoroplatinate, the first noble gas compound to be synthesized. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Xenon is a chemical element with the symbol Xe and atomic number 54. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
